GLP‑1 receptor agonists were developed to mimic endogenous incretin hormones that regulate appetite, insulin secretion, and gastric emptying. Their ability to produce double‑digit percentage weight loss has led to widespread adoption in obesity management. However, public attention, celebrity endorsements, and online wellness clinics have propelled these drugs into the cosmetic sphere, where they are sought for relatively modest weight reduction rather than for treatment of obesity‑related disease. According to a Google trends analysis, the relative search volume (RSV) in “Ozempic” grew exponentially, at a rate of y = (2 × 10–36)e0.0019x. At the start of the 5-year period, the RSV remained steady from March 2018 until a small peak in June 2021, plateauing again until December 2021. Subsequently, the RSV increased, at first slowly until October 2022, then significantly from December 2022 onwards (Figure 1).1,2
This shift has blurred the line between medical necessity and aesthetic desire, prompting debate about safety, fairness, and appropriate prescribing.
Regulatory agencies such as the US Food and Drug Administration approve GLP‑1 medications for chronic weight management only in:3
•Adults with BMI ≥ 30 kg/m², or
•Adults with BMI ≥ 27 kg/m² plus at least one weight‑related comorbidity (e.g., hypertension, type 2 diabetes, dyslipidaemia).
Use in individuals with BMI < 27 kg/m² does not meet approved criteria and is therefore off‑label. While off‑label prescribing is legal, it relies on clinical judgment rather than robust evidence in this population. Importantly, safety and long‑term benefit data for normal‑weight or mildly overweight individuals remain limited.4
One of the strongest drivers of cosmetic GLP‑1 use is social influence. Social media platforms amplify before‑and‑after transformations, often without disclosure of side effects or the need for ongoing therapy. Peer pressure—particularly in image‑focused professions and social circles—normalises pharmacologic weight loss even when health risks are rare but serious.2,5
Studies of off‑label GLP‑1 use in aesthetic and plastic surgery settings suggest that some patients pursue these drugs primarily to conform to perceived beauty standards rather than to improve metabolic health. This environment may undermine informed consent and inflate expectations of effortless, risk‑free weight loss.5
Common Adverse Effects
The most frequent side effects of GLP‑1 receptor agonists are gastrointestinal, including nausea, vomiting, diarrhoea, constipation, and abdominal discomfort. These effects are dose‑dependent and may limit adherence.6
Serious and Emerging Risks
Although uncommon, more serious adverse events have been reported:
•Pancreatitis (including rare fatal cases)7
•Gallbladder disease, partly related to rapid weight loss8
•Gastroparesis and bowel obstruction, particularly with higher doses or prolonged use9
Concerns also exist about loss of lean muscle mass, nutritional deficiencies, and weight regain after discontinuation, especially when lifestyle interventions are not well integrated.10
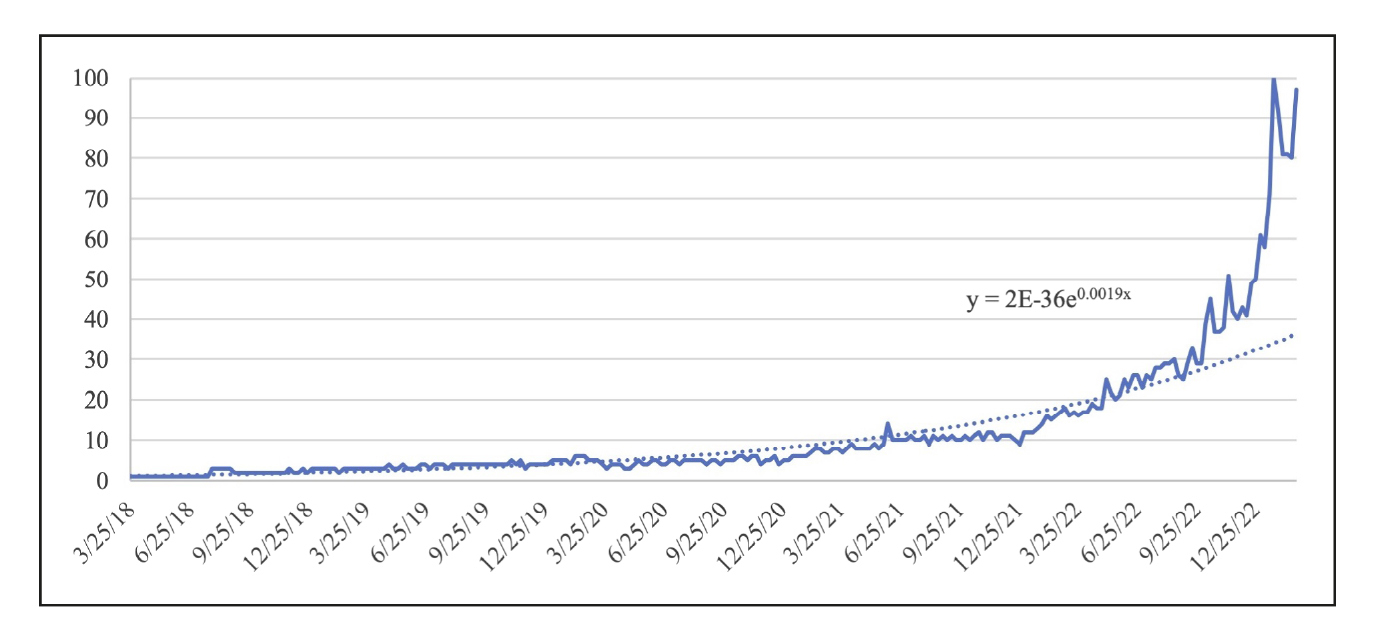
Figure 1. Relative search volume of the term “Ozempic”—March 2018 to February 20232
Risk Amplification in Low‑BMI Individuals
For individuals without obesity‑related disease, the absolute health benefit is smaller, while exposure to pharmacologic risk remains. This alters the benefit–risk equation, making even low‑probability adverse events more ethically significant.11
GLP‑1 medications can produce meaningful short‑term weight loss and appetite control, even in non‑diabetic populations. Some users report improved body image and psychological well‑being. However, these benefits are largely subjective and depend on continued treatment; cessation often leads to weight regain.4
Critically, there is little evidence that cosmetic use confers long‑term health advantages in people without obesity or metabolic risk factors.
Ethical analyses emphasise concerns about:1,12
•Medicalisation of normal weight variation
•Inequitable access, where individuals with medical need face drug shortages
•Commercial influences overshadowing evidence‑based care
Clinicians are encouraged to prioritise medical necessity, conduct thorough risk assessments, and provide transparent counselling about uncertainties and alternatives. Patients, in turn, should critically evaluate whether pharmacologic weight loss aligns with their long‑term health goals rather than short‑term aesthetic pressures.
For patients with obesity and related diseases, the benefits of GLP‑1 therapy clearly outweigh the risks. In contrast, for individuals with BMI < 27 using these drugs for cosmetic purposes, the balance is far less favourable. The potential harms—however rare—may outweigh modest aesthetic benefits, especially when non‑pharmacologic approaches could achieve similar outcomes with fewer risks.1,11
GLP‑1 receptor agonists represent a major advance in obesity medicine, but their growing use for cosmetic weight loss highlights the tension between medical innovation and societal pressure. Off‑label use in individuals with BMI < 27 occurs in a context of limited evidence, potential health risks, and powerful social influences. A cautious, ethically grounded approach—focused on informed consent, individualised risk assessment, and prioritisation of medical need—is essential. Ultimately, sustainable health should outweigh transient aesthetic ideals.